
Preeclampsia Foundation Leads Effort to Mobilize Home Blood Pressure Cuffs
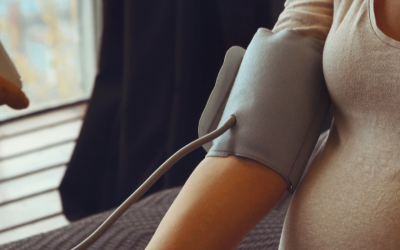
 The Preeclampsia Foundation is responding to the needs of pregnant and postpartum women, during COVID-19, with the introduction of the Cuff Kit®. With women’s obstetric care being monitored by telehealth or otherwise impacted by restrictions and access to care, the Foundation is partnering with healthcare leaders and funders to equip women with home blood pressure monitoring devices and educational material.
The Preeclampsia Foundation is responding to the needs of pregnant and postpartum women, during COVID-19, with the introduction of the Cuff Kit®. With women’s obstetric care being monitored by telehealth or otherwise impacted by restrictions and access to care, the Foundation is partnering with healthcare leaders and funders to equip women with home blood pressure monitoring devices and educational material.
As many as one in twelve pregnancies is affected by hypertension in its most dangerous form –preeclampsia. The Foundation recommends keeping up with prenatal and postpartum visits that include blood pressure monitoring and a review of the signs and symptoms for preeclampsia.
“With COVID-19, we know some prenatal and postpartum appointments have become virtual, and women are reluctant to physically enter healthcare environments” says Eleni Tsigas, Chief Executive Officer. “We had to do something. Missing dangerously high blood pressure will be disastrous for moms and their unborn babies.”
The Foundation convened funding partners, sourced various manufacturers of validated blood pressure devices, and brought patient partners to the table to produce and launch the Cuff Kit®. For women who own a blood pressure monitor, instructional material is free at www.preeclampsia.org/blood-pressure. There, pregnant and new moms can download blank logs or learn about blood pressure tracking apps, watch video tutorials, and receive other information related to blood pressure and their health.
It started with a collaboration with University of Chicago and quickly grew. The first shipments are headed to Nebraska where there’s been a surge in COVID cases in communities with meat packing plants and to several of our nation’s hot spots such as New York City, New Orleans, select counties in Pennsylvania, and the Navajo nation in southwestern states.
The Foundation’s medical experts agree that telehealth will become more commonplace for certain aspects of prenatal and postpartum care, even beyond the current pandemic.
Home monitoring can mean the difference between life and death for women like Whitney Williams of Lexington, KY, who at 34 weeks pregnant was monitoring her blood pressure at home.
When the reading displayed 140/100, she called her doctor who acted promptly. The next 48 hours were a whirlwind, her liver enzymes reached critical levels, her baby was delivered 6 weeks premature, and she ended up having an emergency hysterectomy.
Though Whitney’s case may sound extreme, preeclampsia is not rare - it strikes without warning typically between 20 weeks gestation and up to six weeks after delivery. Whitney and her baby survived, but had she not been checking her blood pressure and acted upon her reading this story could have had a different ending.
For information about the Cuff Kit® or to become a funding partner, please fill out our funding interest form or call 321-421-6957.
###
About the Preeclampsia Foundation
The Preeclampsia Foundation is a U.S.-based 501(c)(3) non-profit organization established in 2000 to improve the outcomes of hypertensive disorders of pregnancy by educating, supporting and engaging the community, improving healthcare practices, and finding a cure. We envision a world where preeclampsia and related hypertensive disorders of pregnancy no longer threaten the lives of mothers and babies. For more information, visit www.preeclampsia.org.
Related Articles

Congratulations on receiving your brand new Cuff Kit®! Want to learn more about how to use your iHealth Track device? Here are some handy videos and links to get you started. Unpacking and using...

Your story is needed to improve outcomes for moms like you. Add your voice to critical preeclampsia research to ensure that every story is heard.

Nurses play a vital role in detecting preeclampsia and caring for patient before, during, and beyond pregnancy.

Doulas can help bridge the gap for any mom, but especially those most vulnerable to maternal illness and death.
The Preeclampsia Foundation announced the launch of a new national initiative to strengthen risk identification through a standardized wristband to help medical teams quickly recognize postpartum comp...

Preventing and managing high blood pressure with healthy lifestyle behaviors are at the center of updated clinical guidelines published this week in the American Heart Association (AHA) peer-reviewed...
Hypertensive disorders of pregnancy are a leading cause of maternal death in the state of Indiana. To address this critical issue, the Indiana Hospital Association is teaming up with the Preeclampsia...

Recientemente, me encontré con una publicación en las redes sociales señalando la crisis de salud maternal desde la perspectiva de una mujer negra. Una persona respondió a...

For more on the Preeclampsia Foundation's work to amplify all research related to biomarkers for improved prediction and diagnostic tools, please visit https://preeclampsia.org/biomarkers. INDIANAPOL...

